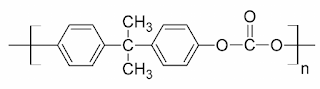
Types of Rubber, their Properties and Applications
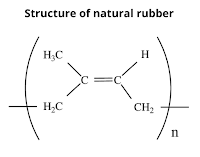
1. Natural Rubber Natural rubber is a natural polymer that is manufactured from latex which is a colloidal solution of rubber particles in water. Latex is obtained by making cuts in the bark of rubber trees like Hevea brasiliensis, found in tropical and semi-tropical countries such as southern. India, Indonesia, Malaysia, Sri Lanka, South America, etc. The natural rubber has remarkable elasticity and undergoes long-range reversible extension even under a relatively small applied force. Preparation of natural rubber Natural rubber is a linear 1,4-addition polymer of isoprene. Since each repeating unit in polyisoprene contains a double bond having cis-stereochemistry. That's why natural rubber is cis-polyisoprene. Properties of Natural rubber Natural rubber has no polar groups and hence intermolecular forces of attraction are only weak van der Waals interactions. Cis-polyisoprene does not have a straight-chain but has a coiled structure. As a result, it can be stretched like a spri...