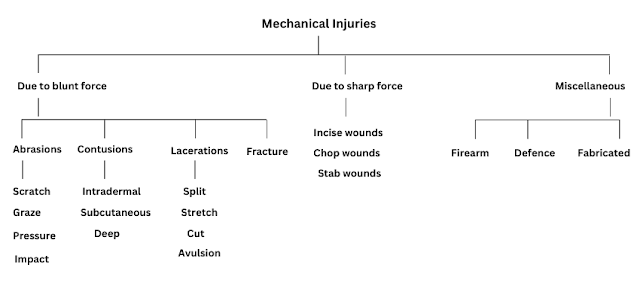
The 12 Principles of Green Chemistry
Green chemistry is the branch of chemistry which aims at the designing of chemical products and processes to reduce or eliminate the generation of hazardous substances.
Green chemistry promotes the green process, a healthy environment, and sustainable development. So, Green chemistry works on some set of principles called "12 Principles of green chemistry". These principles were formulated by Paul T. Anastas.
12 principles of green chemistry are given below:
1. Prevention of waste or by-products
This principle of green chemistry states that we should minimise or eliminate waste by-products rather than cleaning the waste after it has been created.
For example, the production of plastic by-products should be minimised instead of cleaning it after its production. We should make a proper plan to minimise the waste at every step of the chemical process.
2. Maximum incorporation of the reactants (starting materials and reagents) into the final product
This principle of green chemistry is also known as the atom economy. This principle states that green methods should be designed to maximise the incorporation of all materials used in the process into the final product. In other words, we should take the maximum economy from each atom of reactants so that our yield is high.
3. Prevention or minimisation of hazardous products
The most important principle of green chemistry is to prevent or at least minimise the formation of hazardous products, which may be toxic or environmentally harmful. This can be done by introducing a green chemical reaction and synthesis.
4. Designing safer chemicals
This principle of green chemistry states that all the chemicals synthesised or developed (like dyes, paints, cosmetics, etc) should be safe to use for mankind.
For example, Use of thalidomide in pregnant women for lessening the effects of nausea was banned due to birth defects in a child. So, it is very important to design green chemicals that have no side-effect on humans.
This can be done by introducing a new manufacturing process, technology, and manipulating the molecular structure of the chemicals.
5. Energy requirement for synthesis
This principle of green chemistry states that in any chemical synthesis, the energy requirement should be kept minimum.
For example, Certain chemical reaction needs heating, cooling, etc. So, there will be an extra need for energy which gives an unnecessary burden on the energy source.
So, to keep energy requirement at a minimum level, we should use new green manufacturing or synthesis in which there is no need for purification of a compound, supplying extra heat, cooling of chemicals, etc due to which synthesis will be efficient and use minimum energy.
6. Selection of Appropriate solvent
This principle of green chemistry states that the solvent selected for a particular reaction should not cause any environmental pollution and health hazard.
For example, Benzene is used as a solvent in many industrial processes which is toxic and carcinogenic. This problem can be solved by introducing aqueous phase solvents or liquid carbon dioxide as a solvent. Another problem with traditional solvents is that they are volatile and may damage human health and the environment.
This problem can be solved using an immobilised solvent which maintains the solvency of the material and safe for humans and the environment.
7. Selection of starting material
This principle of green chemistry states that we should use renewable starting material instead of non-renewable material.
For example, Use of petrochemicals as starting material during a synthesis should be avoided as they are non-renewable and takes millions of years to the formation. This problem can be solved by introducing green starting materials obtained from agricultural and biological products. They are renewable and good for the environment.
8. Use of Protecting groups
This principle of green chemistry states that if an organic molecule contains two reactive groups and we want to use only one of these groups, the other group has to be protected, the desired reaction completed and the protecting group removed.
For example
This reaction is very common for the synthesis of pesticides. In the above protection, benzyl chloride (a hazard) and the waste generated after deprotection should be handled carefully.
The main limitation in this principle of "using of protecting groups" is that protected groups or reactants are not involved in a reaction, thus making the reaction less atom-economical. So, the use of a protective group should be avoided whenever possible.
9. Use of catalyst
This principle of green chemistry states that we should use a catalyst in a reaction whenever possible because it can increase or decrease the rate of reaction without involving itself in the reaction. This makes a reaction more economical.
Following are the advantages of using a catalyst during a reaction:
1. Better yields
Catalyst helps to obtain better yields during a synthesis.
2. Feasibility of reaction
By using a catalyst, the reaction becomes feasible in those cases where no reaction is normally possible.
3. Selectivity enhancement
By using a catalyst, the reaction will be more selective. Thus the formation of necessary by-products will not take place.
Other advantages:
1. Better utilisation of starting materials.
2. Minimum waste product formation.
3. Minimum external energy requirement.
10. Products designed should be biodegradable
This principle of green chemistry states that products obtained from chemical reaction or synthesis must be biodegradable.
For example, there are three types of pesticides i.e organophosphates, carbamates, and organochlorides in which organophosphates and carbamates are more biodegradable as compared to organochlorides. Organochlorides lead to many problems like biomagnification, decrease in the animal population, loss of fertility of the soil, etc.
Thus, the product must be biodegradable.
11. Designing of manufacturing plants
This principle of green chemistry states that manufacturing plants should be designed in such a way that it eliminates the possibility of an accident during operation.
Accidents like the Bhopal gas tragedy, the Flixborough disaster, and the LG polymer gas leak could be avoided if manufacturing plants were using green synthesis, located in a remote area and following all rules, and regulations. Thus, Green chemistry can minimise the possibility of such disasters.
12. Strengthening of analytical techniques
This principle of green chemistry states that analytical techniques should be so designed that they require minimum usage of chemicals, like recycling of some unreacted reagent for the completion of a particular reaction.
Further, the placement of accurate sensors to monitor the generation of hazardous by-products during a chemical reaction is also advantageous.