Flame Emission Spectroscopy: Instrumentation, Working and Applications
What is flame emission spectroscopy?
Flame emission spectroscopy provides us atomic emission spectrum based on the excitation of atoms from a lower energy state to a higher state. When these atoms return to their original state, they emit radiations which are used to draw the graph.
This technique is widely used to determine the presence of unknown elements like alkali and alkali earth metals in the sample. This is supported by the fact that different metals have different band gaps and they will show different emission spectrums.
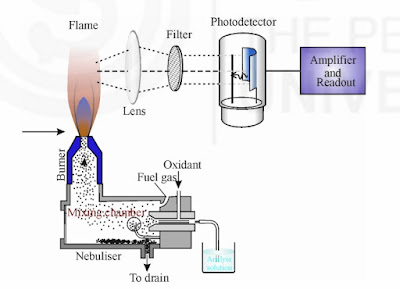
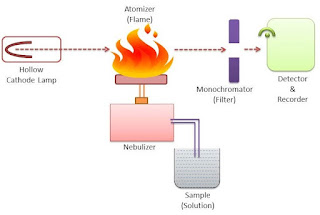
Instrumentation of flame emission spectroscopy
1. Sample
The sample is kept in a sample chamber which is connected to a tube with a nebulizer. The sample may be liquid or solid. The solid sample is first dissolved in a solvent and then analyzed. The sample cannot be a gas.
2. Nebulizer
The nebulizer is a closed chamber connected with the sample through a tube. It also has a continuous supply of fuel and oxidants for the smooth flow of the flame. The nebulizer also contains inert gas which acts as a carrier of the sample molecules toward the flame.
3. Burner/Flame
The burner has a continuous glowing flame that is connected to the nebulizer. The flame is continuously glowing through the continuous flow of fuel and oxidants.
It is on the flame in which the atomization of the sample molecule takes place that the atoms of the molecules gas from the lower energy level to the higher energy level and then return to the ground state by emitting the radiation of a suitable wavelength.
4. Lens
The lens is used to converge the emitted radiations into a single path. This is done to avoid the loss of energy from the emitted radiation. This will improve the accuracy of the graph.
5. Monochromator
The monochromator helps to separate the wavelengths of the emitted radiation. A monochromator is adjusted with the help of computer software.
6. Detector
The detector helps to detect the emitted radiation that is passed through the monochromator. A good detector should be able to detect even a weak signal to produce better accuracy.
7. Computer
A computer is an integral part of the flame emission spectrometer. It helps us to draw the graph of the emission spectrum for various atoms of the sample. It is done with the help of suitable software.
Working of flame emission spectrometer
1. Desolvation
The sample is mixed in the solvent in the sample chamber. So, in this step, the metal particles are dehydrated in the flame thus evaporating the solvent. Now only metal particle remains in the flame.
2. Vaporisation
The metal particles are further dehydrated and they get converted into gaseous particles. That's why this process is known as vaporization.
3. Atomization
This is the most crucial step. In this step, the metal ions or molecules get bifurcated into an individual atom with the application of extreme temperature.
Different metals have different atomization temperatures, some of them require only less temperature whereas some of them require temperature up to 1000 ℃.
4. Excitation
In this step, the atoms absorb energy from the flame and get transferred from the lower energy state to the higher energy state. The excitation energy will be different for different metals as different elements have different band gaps.
5. Emission
Finally, when the atoms return to their ground state they emit the absorbed radiation. The emission radiation has different wavelengths by different elements.
This is finally passed through a monochromator and detector and a graph or emission spectrum is drawn. The emission spectrum will be different for different metals.
Application of flame emission spectroscopy
- It is used for the analysis of ferrous and non-ferrous alloys.
- It is used for the determination of trace metal impurities in alloys, metals, reagents, and solvents.
- It is used for the analysis of metals in geological, environmental, and biological materials.
- Water analysis ICP instruments have been coupled with mass spectrometers to provide a powerful analytical technique.