Diagnosis and Management of Poisoning Cases
Diagnosis of Poisoning Cases
1. Diagnosis of poisoning in living
Following are the symptoms that suggest the poisoning in living:
- The sudden onset of symptoms such as abdominal pain, nausea, vomiting, diarrhea, and collapse.
- The sudden occurrence of coma accompanied by pupil constriction.
- Unexplained coma, especially in children.
- An adult with a known history of depressive illness experiencing a state of coma.
- Rapid onset of neurological or GIT illness.
- Sudden onset of convulsion.
- Delirium with dilated pupils.
- Paralysis, Jaundice, and hepato-cellular failure.
2. Diagnosis of poisoning in the dead (Postmortem Examination)
Internal Findings
1. Smell
To detect unusual odors in the brain, the first step is to open the skull.
2. Mouth and throat
Thoroughly examine for indications of inflammation, erosion, or staining. Necrosis of the pharynx will be seen in case of death caused by sulphonamide and barbiturates.
3. Respiratory System
Corrosive poisons show edema and congestion of the mucous membrane of the trachea and bronchi.
4. Esophagus
The mucous membrane experiences significant softening and desquamation when exposed to corrosive alkalis.
5. Heart
Poisoning with arsenic, antimony, barium, and mercury can lead to the occurrence of subendocardial hemorrhage (internal bleeding) in the left ventricle.
6. Stomach
- Hypermia of muscous membrane.
- Redness of mucosa in case of asphyxial deaths.
- Ulceration caused by corrosives or irritants is typically observed at the greater curvature.
- Perforation is observed when strong mineral acids especially H₂SO₄ have been taken.
- Softening of the mucous membrane of the stomach caused by alkali corrosives.
7. Liver
Phosphorus, chloroform, TNT, and CCl₄ are known to cause liver necrosis. Fatty liver is caused by arsenic, CCl₄, and FeSO₄. Jaundice is caused by phosphorous and potassium chlorate.
8. Kidney
Metal and cantharidin poisoning can lead to the manifestation of parenchymatous degenerative changes.
Necrosis of Proximal Convoluted Tubules (PCT) is observed in mercuric chloride, phenol, Lysol, etc.
Management of Poisoning Cases
1. Removal of Unabsorbed Poison
(a) Inhaled Poisons
In case of poisoning by carbon monoxide, automobile exhaust, gas from the septic tank, etc the patient should be immediately removed to fresh air to remove the poisonous gas through the lungs.
(b) Injected Poisons
If the poison has been injected then some unabsorbed poison may be removed by multiple incisions and suction, similar to that commonly advised for snake bites.
(c) Contact Poisons
If the poison is applied to eyes, skin, wound, or inserted, the poison is removed by washing with plain warm water and administration of specific antidotes.
(d) Ingested Poisons
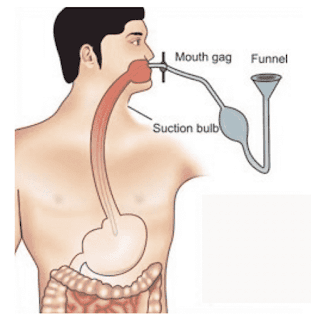
(i) Stomach Washing (Gastric lavage)
Stomach washing is most useful within 1 hr of ingestion of any poison. In this method, 5 ml fluid/Kg of body weight is administered with the help of a french orogastric tube through the mouth.
It is repeated, till the clear and odorless fluid comes out. If any bleeding occurs, the procedure is halted or abandoned.
(ii) Emetics (Early Vomiting)
Early vomiting (emesis) is more effective in preventing absorption than performing vigorous gastric lavage after a delay. Emetics is easier than gastric lavage and less traumatic for the patient.
Emesis should be avoided in corrosive poisoning as it may damage the esophagus and stomach.
Examples of household emetics are copious lukewarm water, and 15 g of mustard powder in 200 ml of water.
(iii) Demulcents and Bulky Foods
These are the substances that prevent the absorption of the poison by forming a coating on the mucous membrane of the stomach. Common demulcents include fats, oils, milk, and egg albumin.
Bulky foods like bananas can serve as a mechanical antidote to glass by trapping its particles and preventing its harmful effects.
4. Administration of Antidotes
Antidotes function by mechanically preventing the action of the poison, without directly destroying or inactivating its harmful effects.
They are also used because the poison may not have been completely removed by emesis or lavage or the poison is already absorbed or it has been administered by other routes than ingestion. Based on their mode of action, antidotes are classified into the following categories:
(i) Physical/ Mechanical Antidotes
Physical antidotes prevent the absorption of poisons in the body by their presence. Following are some physical antidotes:
Activated Charcoal
It is a high-temperature product resulting from the destructive distillation of wood pulp, appearing as a fine, black, and odorless powder.
It is given about 1g/kg of body weight, mixed with water to form a soup-like mixture, and given orally. It acts mechanically by absorbing and retaining the poison within its pores, especially alkaloid poisons.
(iii) Chemical Antidotes
Chemical antidotes work by either forming harmless or insoluble compounds with poisons or by oxidizing the poisons upon contact, thereby preventing their harmful actions. For eg: Potassium permanganate has oxidizing properties.
Its 1:5000 solution is used and the wash must be continued till the solution coming out of the stomach is pink in color. Other examples of chemical antidotes are tannic acid, milk of magnesia, common salt, tincture iodine, etc.
(iii) Physiological/ Pharmacological Antidotes
These agents produce effects that are opposite to that of poison. They are used after some poison is absorbed into circulation. However, the antagonism is usually not complete and the remedy may itself produce undesired results.
Nalorphine has proven to be an effective specific antidote against morphine and its related substances, offering significant value in treatment. Specific chelating agents are commonly employed as antidotes against specific heavy metals, forming stable non-ionized cyclic complexes (Chelates) with cations.