Factors affecting adsorption of gases on solids
What is Adsorption?
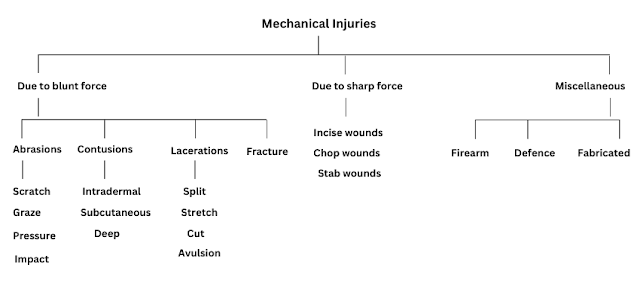
The phenomenon of attracting and retaining the molecules of a substances on the surface of a liquid or a solid resulting into a higher concentration of the molecules on the surface is called adsorption.
The substance thus adsorbed on the surface is called the adsorbate and the substance on which it is adsorbed is called adsorbent.
Factors Affecting Adsorption of Gases on Solids
Almost all solids adsorb gases to some extent. However, the exact amount
of a gas adsorbed depends upon a number of factors as briefly explained
below:
- Nature and Surface area of the adsorbent.
- Nature of the gas being adsorbed.
- Temperature.
- Pressure.
- Activation of solid adsorbent.
1.Nature and Surface area of the adsorbent
It is observed that the same gas is adsorbed to different extents by different solids at the same temperature. Further, as may be expected, the greater the surface area of the adsorbent, greater is the volume of the gas adsorbed. It is for this reason that substances like charcoal and silica gel are excellent adsorbent because they have highly porous structures and hence large surface areas.For the same reason, finely divided substances have larger adsorption power than when they are present in the compact form.
Since the surface area of adsorbents cannot always be determined readily, the common practice is to express the gas adsorbed per gram of the adsorbent ( The surface area per gram of the adsorbent is called specific surface area ).
2.Nature of the gas being adsorbed
Different gases are adsorbed to different extents by the same adsorbent at the same temperature. It may be seen that higher the critical temperature of a gas, greater is the amount of that gas adsorbed. In other words, a gas which is more easily liquefiable or is more soluble in water is more readily adsorbed.This relationship is not surprising as the critical temperature of a gas is related to the intermolecular forces which is an important factor for adsorption too.
Higher the critical temperature, more easily the gas can be liquefied, i.e., greater are the intermolecular forces of attraction. For such a gas, greater will be the attraction of the gas molecules on the surface of the adsorbent and hence greater will be the adsorption.
3.Temperature
Studying the adsorption of any particular gas by some particular adsorbent, it is observed that the adsorption decreases with the increase of the temperature and vice versa. For example, 1 gram of charcoal adsorbs about 10 ml of Nitrogen at 273 K, 20 ml at 244 K and 45 ml at 195 K. The decrease of adsorption with the increase of the temperature may be explained as follows:Like any other equilibrium, adsorption is a process involving a true equilibrium. The two opposing processes involved are condensation ( i.e., adsorption ) of the gas molecules on the surface of the solid and evaporation (i.e., desorption ) of the gas molecules from the surface of the solid into the gaseous phase.
The amount of heat evolved when 1 mole of the gas is adsorbed on the adsorbent is called heat of adsorption.
4.Pressure
At constant temperature, the adsorption of a gas increases with the increase of pressure. It is observed that at low temperature, the adsorption of a gas increases very rapidly as the pressure is increased from small values.5.Activation of solid adsorbent
It means increasing the adsorbing power of an adsorbent. This is usually done by increasing the surface area ( or the specific area ) of the adsorbent which can be achieved in any of the following ways:- By making the surface of the adsorbent rough: e.g., by mechanical rubbing or by chemical reaction or by depositing finely dispersed metals on the surface of the adsorbent by electroplating.
- By subdividing the adsorbent into smaller pieces or grains: No doubt, this method increases the surface area but it has a practical limitation, that is, if the adsorbent is broken into too fine particles that it becomes almost powder, then the penetration of the gas becomes difficult and this will obstruct adsorption.
- By removing the gases already adsorbed, e.g., charcoal is activated by heating in superheated steam or in vacuum at a temperature between 623 and 1273 K.
Cheers
Sciencedoze